Dr. Cristina Florescu Moraid, CEO of AVANTYO Institute of Clinical Research has been invited to open the works of the 9th Regular Conference “Partnership in Clinical Trials”, held in Belgrade, on December 13, 2019 with the presentation "Clinical Trials - Perpetually shifting geographical footprints". The event was organized by CRA Academy from Belgrade.

The geographical distribution of clinical trials is slowly shifting from developed nations to emerging countries. The rising cost of clinical trials and difficulty in patient recruitment has led biopharmaceutical companies to shift towards regions such as Central and Eastern Europe, Asia Pacific, Latin America, and Middle East for cost efficiency and quick patient recruitment.
Emerging countries also possess greater disease variation compared to west, where traditional diseases are growing. The greater disease variation among the developing countries helps biopharmaceutical companies to perform clinical trials from rare diseases and not only.
Biopharmaceutical companies have shift towards regions such as Central and Eastern Europe, Asia Pacific, Latin America, and Middle East for cost efficiency, quick patient recruitment and greater disease variation.
 Interesting findings regarding CEE were mentioned. The CEE is one of the most dynamic and fastest-growing regions in Europe, and there are several advantages to placing clinical research studies in these areas, including:
Interesting findings regarding CEE were mentioned. The CEE is one of the most dynamic and fastest-growing regions in Europe, and there are several advantages to placing clinical research studies in these areas, including:
1 - Rapid recruitment of study population
2 - Investigators are highly motivated and loyal
3 - Remarkable price advantage
4 - Local monitors are more skilled than average
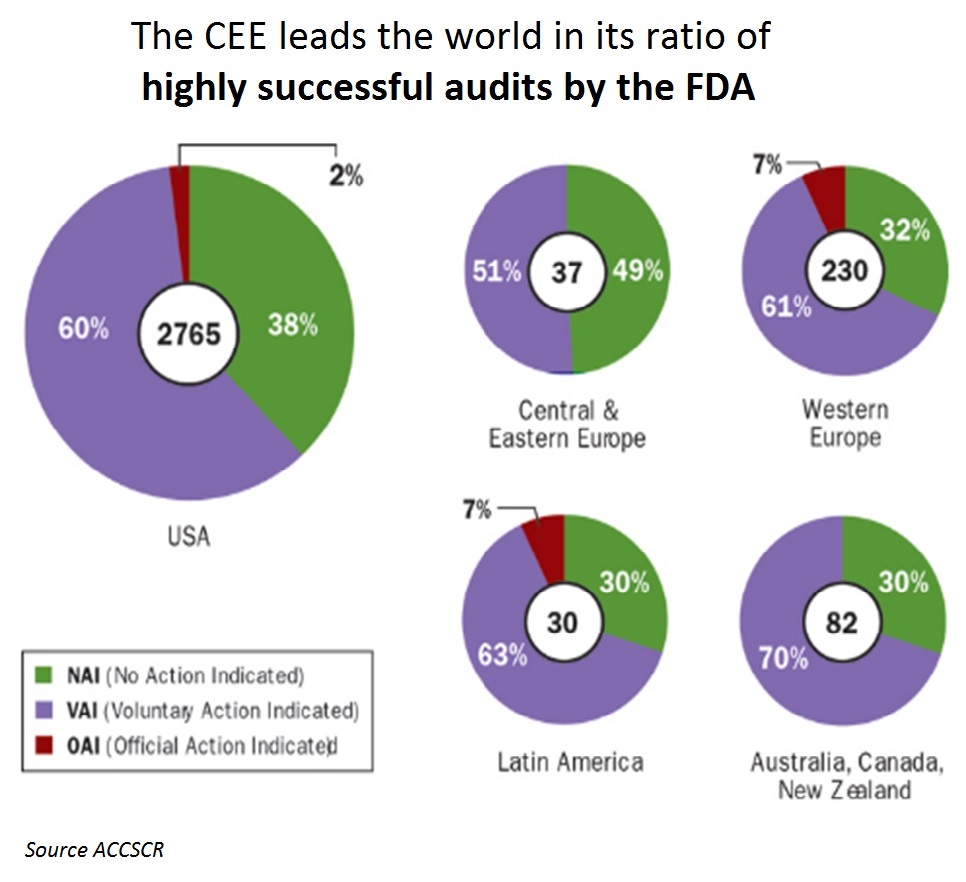
5 – Excellent audit results of USA Food and Drugs Agency (FDA)
Digitization in biomedical research is paving the way for growth of global clinical trials market. Adoption of Systems like EDC also helps companies manage their patient data which ultimately reduces the monitoring cost and better patient compliance. Digitization also helps meet the stringent regulations by maintaining patient data records which ultimately help reduce trial process errors.
Three main conclusions about future of clinical trials:
1. Clinical Trials Will Be Open
Open clinical trials will involve connected infrastructure, consistent operations, and cooperative stakeholder involvement. A system in which trial operations are no longer a “black box”
2. Clinical Trials Will Be Human-Centered
As open clinical trials enable greater focus on science and patient value, rather than operations, industry will further orient itself around the humans served by research. This will be a necessary shift
3. Clinical Trials Will Be More Integrated
As trials become more open and human-centered, society will have improved capacity to better understand and interact with clinical research. Trials can become more integrated in the medical system and perceived as a critical component of advancing health.





